Look, navigating the global medical device supply chain is a massive headache right now. If you’re a regional distributor, an agency owner, or handling procurement for a large healthcare network, you already know the stakes. Picking the wrong supplier for surgical power tools doesn’t just eat into your profit margins—it ruins your reputation with surgeons and hospitals. And in this industry, reputation is literally everything.
We are talking about highly precise, deeply critical equipment here. According to a recent report by Grand View Research, the global arthroscopy devices market was valued at around USD 5.3 billion in 2022 and is growing steadily. The demand is there. Sports injuries are up, aging populations need more joint surgeries, and minimally invasive procedures are the gold standard. But here is the dirty little secret no one talks about at trade shows like MEDICA or Arab Health: most companies claiming to be an arthroscopy shaver system manufacturer are just glorified assembly shops.
They buy cheap motors from one place, generic circuit boards from another, slap a shiny logo on the console, and call themselves a factory. When the handpiece overheats mid-surgery or the autoclavable seals fail after 20 washes, who takes the blame? You do.
So, let’s cut the corporate fluff. I’m going to walk you through exactly how to weed out the pretenders and find a legit medical equipment OEM that can actually support a global distribution business.
The “Fake OEM” Trap and Why It Hurts Your Bottom Line
I’ll just say it—I have a massive issue with how the term “OEM” is thrown around in the medical device world today. A true OEM (Original Equipment Manufacturer) engineers the product, controls the raw materials, writes the software, and manages the quality control of every single component.
A lot of so-called orthopedic surgical instruments supplier companies are really just traders. Why is this a problem for you as a distributor?
Because when a surgeon complains that the shaver blade is clogging during an ACL reconstruction, a fake OEM cant fix the problem. They don’t own the source code for the console’s algorithm, and they don’t machine their own blades. They just pass your complaint down the chain, and you’re left waiting months for a solution while your client buys from a competitor.
If you want long-term commercial success, you need a partner that handles true research and development. You need a factory that can tweak the motor torque curves if your local market demands it.
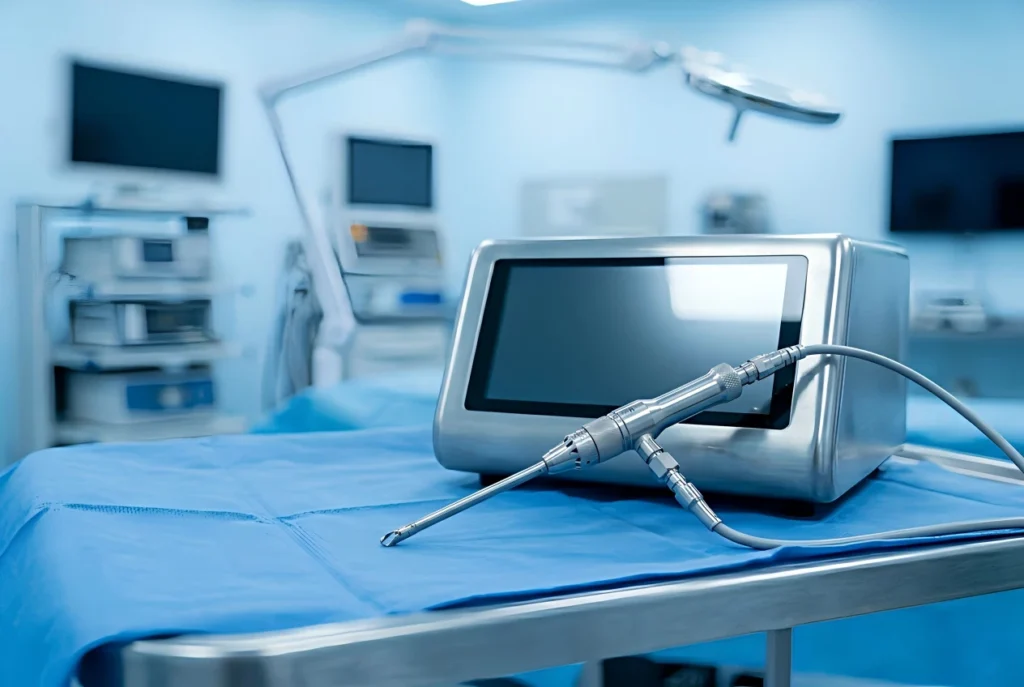
Arthroscopy Shaver System for Orthopedic Joint Surgery | High-Speed Endoscopic Shaver for Knee and Shoulder Procedures | OrthoPro Surgical Power Tools
The Arthroscopy Shaver System by OrthoPro is an advanced surgical solution designed for precision soft tissue resection and bone debridement. This high-performance Arthroscopy Shaver System offers surgeons exceptional control and efficiency during minimally invasive joint surgeries. Engineered for durability, our Arthroscopy Shaver System ensures optimal clinical outcomes in knee, shoulder, and hip arthroscopy.
Non-Negotiable Criteria for Your Next Supplier
When you’re looking to import wholesale arthroscopy tools in bulk, you need to audit the factory brutally. Don’t just look at their glossy brochures. Here is what actually matters.
1. Certifications That Aren’t Just Window Dressing
Everybody says they have CE and ISO. Big deal.
With the shift from MDD to MDR in Europe, the regulatory landscape has become a nightmare. Getting an MDR CE mark is expensive and rigorous. If a factory is still quoting outdated MDD certificates or trying to pass off a “Declaration of Conformity” for a Class IIa or IIb device without a Notified Body number, run the other way.
You need ISO 13485:2016, specifically for the design and manufaturing of surgical power tools. Ask to see their last audit report. If they hesitate, it means their Quality Management System (QMS) is a mess, and your shipments are going to get stuck at customs.
2. Stable Production Capacity for Large Tenders
Winning a government hospital tender is an amazing feeling right up until your supplier tells you the lead time is 180 days because they ran out of micro-motors.
A reliable arthroscopy shaver system manufacturer should have scalable production lines. Ask them point-blank: “What is your maximum monthly output for shaver consoles and handpieces, and what is your current capacity utilization?” If they are running at 99% capacity, they have no room for your urgent orders.
3. Deep OEM and Customization Capabilities
You aren’t just buying boxes; you are building a brand. Whether you want to white-label the equipment or need specific UI modifications on the touchscreen console to support local languages (like Spanish, Arabic, or Portuguese), the factory needs an in-house software team. Can they laser-engrave your logo on the handpiece? Can they customize the footswitch layout? These details matter to end-users.
Technical Specs You Absolutely Cannot Compromise On
Surgeons are picky, as they should be. When evaluating an Arthroscopy Shaver System, you need to understand the physics of what makes a good machine. If you don’t know the tech, the hospital procurement committee will eat you alive.
Motor Performance: Speed vs. Torque
Arthroscopy shavers need to handle different tissues. Soft tissue (like meniscus) requires high speed and sharp cutting, while bone resection (like acromioplasty) requires lower speed but massive torque.
Usually, a top-tier system will offer:
- Oscillation modes up to 3,000 – 4,000 RPM
- Forward/Reverse continuous modes up to 8,000 RPM
But speed means nothing without torque. If the torque is weak, the blade stalls when it hits dense bone.
Here is a simplified way we look at cutting performance in the industry (no fancy math blocks, just plain text):
Effective Resection Rate (ERR) = (RPM * Blade Window Area * Torque Factor) / Tissue Density Resistance
If your supplier’s motor lacks the Torque Factor, the ERR drops to zero the second the surgeon applies pressure. The motor burns out, the handpiece gets hot, and the surgery is delayed. Always ask the supplier for their torque specifications under load.
Fluid Ingress and Autoclavability
The number one reason arthroscopy handpieces fail is fluid ingress. Saline gets inside the motor housing, and it shorts out.
A premium handpiece must be rated IPX8. Furthermore, it must survive hundreds of sterilization cycles. The standard hospital autoclave cycle is 134 degrees Celsius (273F) at high pressure for up to 18 minutes. Cheap plastics and inferior O-rings will crack after 20 cycles. You need to ask the factory what grade of materials they use for internal sealing.
The Console and User Interface
The console should automatically recognize the handpiece and the blade type attached. Surgeons dont have time to manually input settings while a patient is under anesthesia. The UI should be intuitive, and the screen needs to be responsive even if the nurse is wearing double sterile gloves.
Evaluating a Supplier: The Cheat Sheet
To make things easier, I put together this quick matrix. I use this exactly when I’m auditing a new orthopedic surgical instruments supplier for a distribution network.
| Evaluation Area | The “Pretender” (Red Flags) | The “Real Deal” (Green Flags) |
| R&D Capability | Outsources PCB design; no software engineers on staff. | In-house R&D team; holds original patents for motor control. |
| Material Sourcing | Buys generic stainless steel; refuses to name bearing suppliers. | Uses medical-grade 440C/316L steel; uses imported high-end bearings. |
| Sterilization Testing | “Calculated” lifecycle based on component specs. | Actual autoclave stress-testing logs showing 500+ cycles. |
| Quality Control | Only spot-checks finished goods before shipping. | Tracks individual components with serial numbers; strict QA at every assembly stage. |
| After-Sales | Requires you to ship broken units back to Asia at your expense. | Provides modular spare parts, video repair guides, and a generous warranty buffer. |
Real-World Case: Surviving the Margin Crunch
Let me share a quick story (keeping names anonymous for obvious reasons). A mid-sized distributor in Latin America was buying shaver systems from a very cheap supplier they found online. Their initial profit margins looked incredible on a spreadsheet.
Six months in, the nightmare started. Handpieces were stalling. The error codes on the consoles made no sense, and the manual was poorly translated English. The distributor had an RMA (Return Merchandise Authorization) rate of nearly 18%. They were losing money on airfreight trying to send units back for repair, and two major private hospitals threatened to cancel all their contracts.
They finally wised up and switched to a legitimate source—one that actually mirrors the standards we uphold at OrthoPro. The unit cost was about 15% higher initially. But here is the kicker: their RMA rate dropped to 0.5%. They didn’t have to keep a massive inventory of loaner units, their sales reps stopped wasting time apologizing to surgeons, and their net profit at the end of the year was actually higher despite the higher upfront cost.
When you buy wholesale arthroscopy tools, you are not just buying metal and wires. You are buying the absence of problems.
Total Cost of Ownership (TCO) for Distributors
You have to think about TCO. Your actual cost isn’t the FOB price on the proforma invoice.
True Cost = (Unit Price) + (Import Duties) + (Cost of Warranty Replacements) + (Lost Revenue from Damaged Reputation)
If a cheap shaver costs you $1,500 but fails in three months, and a high-quality OEM shaver costs $2,200 but lasts five years, which one is actually cheaper? Your hospital clients understand TCO. You need to understand it too when picking your factory.
Why the Right Partner Makes All the Difference
You need a factory that treats you like a partner, not an ATM. A solid manufacturer provides extensive technical training for your sales team. If your reps don’t know how to explain the advantages of a brushless, sensorless motor to a senior orthopedic surgeon, they wont close the deal.
The manufacturer should provide high-res marketing materials, clear technical dossiers for your local Ministry of Health registrations, and fast responses when you have technical queries in different time zones.
This is exactly why so many international distributors are realigning their supply chains and looking for reliable brands that can offer true OEM support, stable pricing, and uncompromised quality.
Frequently Asked Questions (FAQ)
1. How long does it typically take to customize and OEM an arthroscopy shaver system?
If you are just doing basic white-labeling (logo engraving, custom software splash screen, packaging), a capable factory can usually handle this within 30 to 45 days. If you need structural changes to the handpiece or custom blade designs, you are looking at 3 to 6 months for engineering, prototyping, and validation.
2. What is the standard warranty I should expect from a reliable medical equipment OEM?
For the main console and footswitch, 12 to 24 months is industry standard. Handpieces are trickier because they endure harsh sterilization, but a confident manufacturer should offer at least 6 to 12 months on the handpiece, provided the hospital follows proper cleaning protocols.
3. Do manufacturers provide CE/ISO documents for local Ministry of Health registration?
Yes, absolutely. A legitimate manufacturer will supply a complete technical file, including Free Sale Certificates (FSC), ISO 13485, CE certificates, and clinical evaluation reports required by your local regulatory bodies (like ANVISA, COFEPRIS, or FDA). If they stall on providing these, they probably don’t have them.
Let’s Build a Profitable Partnership
If you are tired of dealing with middle-men, inconsistent quality, and poor communication, it’s time to work with a team that actually understands the B2B medical device market.
At OrthoPro, we don’t just sell equipment; we build long-term distribution networks based on trust, elite engineering, and rock-solid after-sales support. Whether you need a reliable brand to take to your local hospitals or a discreet OEM partner to build your own product line, we have the manufacturing muscle and the regulatory backbone to make it happen.
Stop risking your reputation on unverified suppliers. Check out our high-performance Arthroscopy Shaver Systems and see the difference quality makes.
Ready to talk margins, exclusivity, and technical specs? Jump over to our Contact Us page or shoot an email straight to our global team at info@orthopro.mx. Let’s get your supply chain secured.